Landmark CGH surgical trial explores new treatment approach for persons living with Alzheimer’s disease and their families
CGH is the first public hospital to research neurolymphatic surgery to manage the progression of Alzheimer’s disease

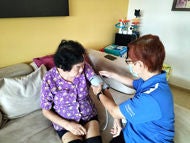
Clin Asst Prof Vincent Tay (left) and Clin Asst Prof Jeremy Sun (right) performing the Cervical Lymphatico-Venous Bypass on a patient with Alzheimer's disease at Changi General Hospital, a procedure aimed to enhance the clearance of neurotoxins in the brain.
Singapore, 7 April 2026 – Changi General Hospital (CGH) has embarked on a surgical trial known as Cervical Lymphatico-Venous Bypass for Alzheimer's Disease (CLyVeB-AD-1), the first regulated clinical trial of its kind in Singapore and Southeast Asia.
The surgical intervention repurposes lymphatic bypass surgery to enhance clearance of neurotoxic proteins in the glymphatic systems of patients with mild to moderate Alzheimer’s disease. This offers a new treatment option that reduces toxic protein biomarkers in the brain, potentially improving behavioural and psychological symptoms for these patients.
There is an estimated 100,000 persons living with dementia in Singapore today, and about 70% of them have Alzheimer's disease, a type of dementia that increasingly affects cognitive function over time. Early signs may include difficulty remembering recent conversations or misplacing items, while later stages can involve challenges with language, navigation, problem-solving, activities of daily living and independence, as well as changes in mood or personality. As Alzheimer’s disease progresses, families and caregivers often face increasing challenges in managing symptoms and providing daily care and support, highlighting the importance of innovative therapies that could offer new possibilities for managing the condition.
First regulated trial of its kind in Singapore and Southeast Asia
Clinical Assistant Professor Vincent Tay, Consultant, Plastic, Reconstructive & Aesthetic Surgery Service, Department of Surgery, CGH, and Principal Investigator of the CLyVeB-AD-1 trial, said, “The disease burden of Alzheimer's continues to grow in Singapore as the population ages. Current medical treatments can only slow disease progression to a certain extent, which is why CGH wanted to explore innovative treatment approaches.”
“This procedure was an opportunity to apply our expertise in lymphatic microsurgery to help patients with Alzheimer’s disease who had limited medical treatment options. After studying the procedure and perioperative care approach, CGH embarked on this translational research to investigate if this surgical approach could offer our patients something more – a chance to not just slow the disease progression but also potentially improve their quality of life and ease the caregiving journey for families,” Clin Asst Prof Tay said.
In January 2025, the CLyVeB-AD-1 trial received approval from the Institutional Review Board (IRB), making it the first study of its kind to obtain IRB approval and be prospectively registered on clinicaltrials.gov outside of China (where the surgery was first rolled out). The trial incorporates comprehensive monitoring protocols and a two-year follow-up period for each patient who undergoes the surgery.
New application of proven surgical techniques
The trial involves carrying out lymphaticovenous anastomosis (LVA) surgery on patients with Alzheimer's disease. LVA surgery is primarily used to treat lymphoedema, a condition that causes fluid accumulation and painful swelling of the limbs. By connecting neck lymphatic structures to adjacent veins in the neck area (i.e. cervical lymphaticovenous bypass), the surgery aims to enhance the clearance of neurotoxic proteins such as amyloid and tau from patients’ central nervous systems via the glymphatic and meningeal lymphatic systems.

Changi General Hospital’s CLyVeB-AD-1 surgical technique: Lymph node to venous end-to-side anastomosis (LNVA), a subtype of LVA surgery, is used to enhance the clearance of neurotoxic proteins in the glymphatic systems of Alzheimer's disease patients.
Clinical Assistant Professor Jeremy Sun, Head and Senior Consultant, Plastic, Reconstructive & Aesthetic Surgery Service, Department of Surgery; Director, Lymphoedema Service, CGH, and Co-Investigator of the CLyVeB-AD-1 trial, said, “This procedure represents an innovative application of lymphatic microsurgery techniques that we have refined over years of treating lymphoedema patients at CGH. The surgical principles are similar – creating bypass pathways for fluid drainage – but the target is different. By connecting neck lymphatic structures to veins to enhance the brain's natural waste clearance system, we can potentially help clear the toxic proteins that accumulate in Alzheimer's disease.”
Promising early results from pilot cohort
To date, four patients aged between 61 and 73 years have undergone the procedure, with encouraging preliminary results observed over six months of patient follow-ups. The patients, who had mild to moderate Alzheimer's disease with biomarker confirmation, demonstrated improvements in at least one clinical assessment, with most patients showing improvement across multiple measures. Longer term follow-up is required to understand the longevity of these changes.
Adjunct Associate Professor Lim Si Ching, Senior Consultant, Department of Geriatric Medicine, CGH, and Co-Investigator of the CLyVeB-AD-1 trial, said, "We are observing meaningful improvements in behavioural and psychological symptoms of our patients with dementia after the procedure, which can go a long way in supporting their families and alleviating the demands on caregivers. One patient who had long-standing depression and anxiety seemed to be less anxious and had improved mood after the surgery. Another patient who had challenges with language became more interactive and communicated better after the surgery, much to the family’s delight. These changes, while preliminary, represent hope for families who previously had limited medical treatment options for their loved ones with Alzheimer’s disease.”
A multidisciplinary care team – comprising physiotherapists, dietitians and nurses trained in dementia and person-centred care – works collaboratively with patients and families throughout their treatment journey. This ensures patients are provided with comprehensive support from initial assessment through to pre-operative, post-operative and long-term follow-up care, including person-centred, designed therapies and activities after surgery.
In evaluating results from the surgical trial, assessment methods include cognitive testing, quality of life measures, caregiver burden evaluation, and biomarker analysis. Notably, preliminary biochemical improvements have been observed through reduced toxic protein biomarkers in patients' cerebrospinal fluid, providing objective evidence of the treatment's potential efficacy.
A family spokesperson for a patient said, “We have observed steady improvements in mobility. One noticeable change immediately post-surgery was how they brought up stories from long ago, some that none of us remembered. We observed the disposition becoming more cheerful, and the ability to recall things has seen gradual improvements.”
Clin Asst Prof Tay added, “This trial represents the essence of translational research – taking scientific discoveries about the brain's waste clearance system and rigorously testing them in a clinical setting. Our goal is to bridge the gap between laboratory discoveries and bedside care, contributing to the global understanding of whether surgical approaches can complement existing treatments for neurodegenerative diseases such as Alzheimer's disease.”
END